文章目录
台北 Taipei
TEL:+886-2-2557-5607
台中 Taichung
TEL:+886-4-2320-2793
上海 Shanghai
TEL:+86 21-6090-4391
深圳 Shenzhen
TEL:+86 755-83176807
China License Qualification Certificate
进口化妆品备案凭证
Imported Cosmetics Record Certificate
进口化妆品备案凭证
Imported Cosmetics Record Certificate
对于在中国从事化妆品经营的企业来说,申请”中国大陆化妆品三证“是必要的。
申请大陆化妆品备案凭证许可批件是企业从事在大陆经营化妆品或要在大陆进出口化妆品新原料的企业或个人必须申请的,是每种化妆产品经营销售、进出口的有效凭证。
随着市场机制的改革,市场经济的越来越规范化,法律体制以及化妆品监管体制的越来越健全化,对于想要在大陆从事化妆品经营的企业来说申请大陆化妆品三证是必要,但必须先申办《化妆品备案凭证许可批件》,若没申请化妆品三证在大陆买卖化妆品将寸步难行,还会违反当地法令。

一、化妆品备案凭证简述
- 中国大陆政府2014年起大力整顿网路上与实体店内无批文的进口化妆品销售行为及规范,而中国化妆品市场急速成长,所有要到中国大陆进口销售前必须取得化妆品三证,企业在上海、深圳都需注册备案申请,所有进口的化妆品经行政管理部门审批,审批通过后才可进行正常的报关报检及销售;未经审批的代办进口的化妆品,无法进行正常报关报检,亦不得在中国大陆市场上销售化妆品。
- 现今化妆品在全球都受到消费者的青睐,早期台商在中国大陆从事化妆品进出口买卖的企业,大多数都经由小三通到中国大陆当地进行销售,然而越来越多的在大陆进出口化妆品经营的企业,政府为了保障企业及消费者的权益,中国大陆要求化妆品类商品进口必须经由正式报关方式才可合法上架销售,所以在中国进行进口化妆品经营的企业就需要在申请大陆化妆品三证才符合规定。
- 化妆品的《批件》是针对每一个具体品种的,只要名称或者配方有一点点不一样,就是另一个产品,就需要另行申报一个《批件》,每个《批件》上都有唯一的一个批准文号,一个批准文号对应一个具体的产品。比如:同一乳液品牌5种颜色,即使基础配方都一样,就是著色剂不一样,按规定要申报5个《批件》;再比如:同一香水品牌8种味道也要申报8个《批件》。另外需要注意的就是中国官方目前没有“药妆”这一类,也不能申报专门的“药妆”类批文。
- 汇佳管理咨询有限公司以多年工商经验专业办理各种中国许可资质证申请,而在中国大陆申请化妆品备案较为严格,汇佳有实质办理化妆品三证申请代办的经验,且从工商登记、(中国)许可资质证申请后续会计服务代理,汇佳国际Inter Area 提供中国公司注册一站式服务,并提供专业人员咨询大陆化妆品备案申请及其他公司设立事务,以让投资者能真正于当地营业为目标,并且能设身处地在当地的为客户留心关照各项工商事务,让投资者能放心投资,专注于业务市场开拓!
二、中国大陆化妆品备案申请凭证类别
- 中国大陆化妆品三证申请之中的【化妆品备案凭证许可】是由中华人民共和国国家食品药品监督管理局对国产特殊用途及进口化妆品(包括特殊类和非特殊类)实行申报审核制度。
- 化妆品要申报国家药监局卫生许可批件(或备案凭证),在化妆品备案申报大致区分为三大类,1.国产特殊类、2.进口特殊类、3.进口非特殊类:
国产特殊用途化妆品
领取《国家食品药品监督管理局国产特殊用途化妆品卫生许可批件》
√ 国产化妆品备案凭证许可申请人应是化妆品生产企业。
√ 国产化妆品原料备案凭证许可申请人应是化妆品原料生产企业或化妆品生产企业。
Cosmetic

进口特殊类化妆品
领取《国家食品药品监督管理局进口特殊用途化妆品卫生许可批件》
√ 育发、美乳、烫发、健美、染发、脱毛、除臭、祛斑、防晒…等均属于进口特殊类化妆品。
进口非特殊类化妆品
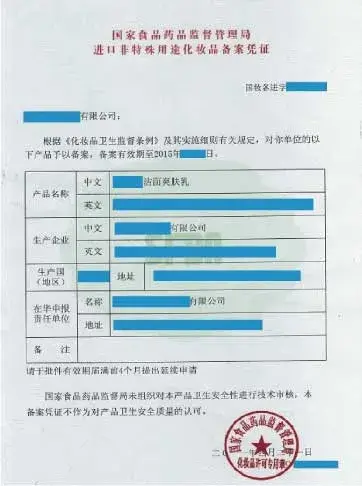
领取《国家食品药品监督管理局进口非特殊用途化妆品备案凭证》
√ 进口化妆品备案凭证许可申请人大多是进口化妆品生产企业。
√ 进口化妆品新原料备案凭证许可申请人大多是进口化妆品新原料或化妆品生产企业。
√ 发用类、护肤类、彩妆类、指(趾)甲类、芳香类…等均属于进口非特殊类化妆品。
三、中国大陆进出口化妆品备案申请的应备文件
I.申请国产特殊用途化妆品行政许可的,应提交下列资料:
- 国产特殊用途化妆品行政许可申请表;
- 产品名称命名依据;
- 产品品质安全控制要求;
- 产品设计包装(含产品标签、产品说明书);
- 经国家食品药品监督管理局认定的许可检验机构出具的检验报告及相关资料;
- 产品中可能存在安全性风险物质的有关安全性评估资料;
- 省级食品药品监督管理部门出具的生产卫生条件审核意见;
- 申请育发、健美、美乳类产品的,应提交功效成份及其使用依据的科学文献资料
- 另附省级食品药品监督管理部门封样并未启封的样品1件;
II.申请进口特殊用途化妆品行政许可的,应提交下列资料:
- 进口特殊用途化妆品行政许可申请表;
- 产品中文名称命名依据;
- 产品配方;
- 生产工艺简述和简图;
- 产品品质安全控制要求;
- 产品原包装(含产品标签、产品说明书);
- 拟专为中国市场设计包装的,需同时提交产品设计包装(含产品标签、产品说明书);
- 经国家食品药品监督管理局认定的许可检验机构出具的检验报告及相关资料;
- 产品中可能存在安全性风险物质的有关安全性评估资料;
- 申请育发、健美、美乳类产品的,应提交功效成份及其使用依据的科学文献资料;
- 已经备案的行政许可在华申报责任单位授权书影本及行政许可在华申报责任单位营业执照影本并加盖公章;
- 申请化妆品使用原料及原料来源符合疯牛病疫区高风险物质禁限用要求的承诺书;
- 产品在生产国(地区)或原产国(地区)生产和销售的证明文件;
- 可能有助于行政许可的其他资料。
- 另附许可检验机构封样并未启封的市售样品1件。
III.申请进口非特殊用途化妆品备案的,应提交下列资料:
- 进口非特殊用途化妆品行政许可申请表;
- 产品中文名称命名依据;
- 产品配方;
- 产品品质安全控制要求;
- 产品原包装(含产品标签、产品说明书);
- 拟专为中国市场设计包装的,需同时提交产品设计包装(含产品标签、产品说明书);
- 经国家食品药品监督管理局认定的许可检验机构出具的检验报告及相关资料;
- 产品中可能存在安全性风险物质的有关安全性评估料;
- 已经备案的行政许可在华申报责任单位授权书影本及行政许可在华申报责任单位营业执照影本并加盖公章;
- 化妆品使用原料及原料来源符合疯牛病疫区高风险物质禁限用要求的承诺书;
- 产品在生产国(地区)或原产国(地区)生产和销售的证明文件;
- 另附许可检验机构封样并未启封的市售样品1件。
IV.备注:
根据国家药监局最新规定,2011年4月1日后化妆品申报材料除了上述资料外,还必须提交”化妆品产品技术要求”以下几种资料。申请人应按照《化妆品产品技术要求编制指南》要求填写化妆品产品技术要求,并符合下列要求:
- 产品名称只需填写产品中文拼音名。
- 配方成分。
- 生产工艺应描述完整的生产工艺,包括主要步骤和工艺流程。
- 感官指标中的颜色描述、性状描述、气味描述。
- 卫生化学指标和微生物指标。
- 化妆品产品技术要求所载明的使用方法和贮存条件应当与化妆品行政许可申报资料中,产品标签或产品说明书注明的使用方法和贮存条件一致。
- 保质期应明确写明该产品的保质期限要求,如保质期××个月(或×年)。
四、中国大陆进口化妆品检验检疫
- 申请化妆品三证之一在进口化妆品通关时,海关要对每个产品逐一核验《批件》,如果没有事先申报《批件》,海关也不会放行。
- 另外,《批件》也是申请进口化妆品在中国大陆合法销售的唯一凭证,正规商场、超市、专卖店进货时都需要您出示每个产品的《批件》,如果没有《批件》商家违规销售也是违法行为。
- 工商、质检、卫生等部门在市场执法检查时,对于没有《批件》的化妆品可以罚没销毁,还要根据情况处以罚金。进口化妆品的收货人或者其代理人应当按照国家质检总局相关规定报检,同时提供收货人备案号。
- 进口化妆品经检验检疫合格的,检验检疫机构出具《入境货物检验检疫证明》,并列明货物的名称、品牌、原产国家(地区)、规格、数/重量、生产批号/生产日期等。进口化妆品取得《入境货物检验检疫证明》后,方可销售、使用。
- 根据《进出口化妆品检验检疫监督管理办法》(国家质检总局令第143号)的规定,自2012年2月1日起,经检验合格的进口化妆品不再加贴检验、检疫标志(CIQ) 。
五、中国大陆进口非特殊用途化妆品备案凭证
六、中国大陆化妆品备案申请流程
- 资料准备。
- 在华申报责人责任单位授权备案。
- 样品鉴验。
- CFDA申请<备案批件>。
- 进口化妆品收货人备案。
七、中国大陆化妆品贸易公司注册–Q&A问答
Q1.何谓中国大陆化妆品三证申请?
A:其实中国大陆俗称的化妆品三证指的是在大陆经营化妆品企业必须申请的【化妆品备案凭证许可】及在大陆进行进出口业务需申请的【海关登记证】、【自理报检企业备案登记证明书】,此进口化妆品需要的三个证书简称「化妆品三证」。汇佳国际具有专业人才,了解客户企业需求,而近几年中国大陆为了简化审批程式,推出三证合一,避免繁琐作业流程。而汇佳国际具有在大陆实质的申请化妆品备案的经验,能为企业节省时间成本,帮客户办理大陆化妆品三证申请,以利客户在大陆从事化妆品业的经营。
Q2.原本从小三通进口到中国大陆的化妆品现在卡关了怎么办?
A:原本中国大陆经由小三通进口的化妆品会卡关,主要原因是大陆政府都希望回归到正常报关程序,因此要在中国大陆经营化妆品贩售的企业,必须经由正常报关程序才可将商品进口到大陆境内销售。 【小三通资讯】
Q3.在中国大陆申请化妆品凭证备案需要多久?
A:在中国大陆进口单一支化妆品就需要做一份化妆品备案,且需要8个月的样品检测,检测合格后才会出具检测报告,而同款化妆品只要是成份甚至颜色不同,就算另一支产品,需在另申请一份化妆品备案。
Q4.什么是在华责任单位?
A:出口到中国大陆的每支化妆品只能授权给一个代理单位,而代理单位必须在中国大陆当地具有独立法人资格的企业,其在华责任单位指的就是中国大陆的代理企业;代理在中国大陆进口化妆品时需要提交化妆品进口产品成的分进行单位检测,意味着代理企业的选择势必是投资者自己成立的企业亦或是中国大陆的合作企业。